  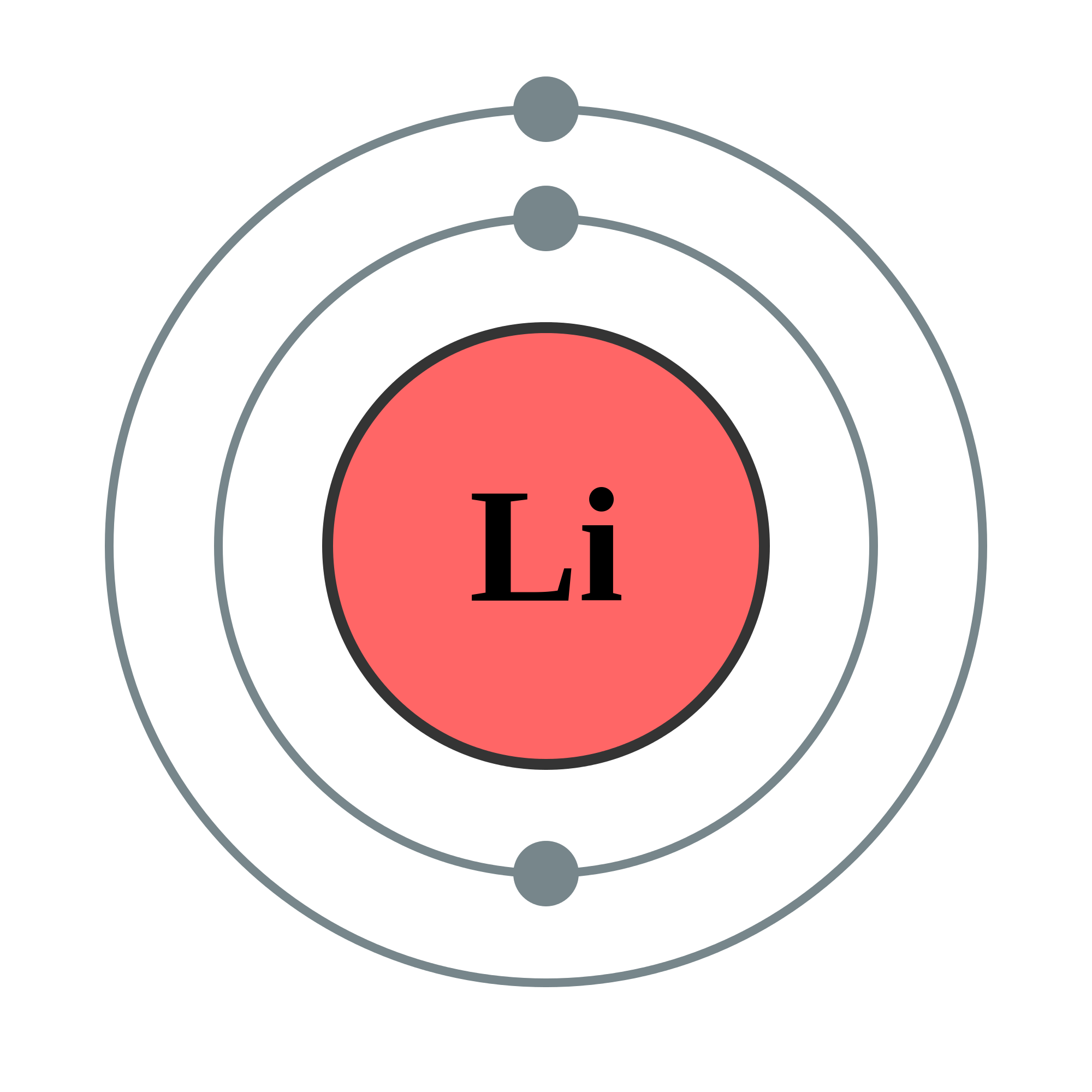
The outermost energy level or shell of an atom is known as the valence shell. The electrons are allowed to jump from lower to higher energy levels as they absorb energy and also, drop down from higher to lower energy levels as they release energy. The electrons located in the shell closest to the nucleus have minimum energy and are said to be in the ground state. The number increases away from the nucleus as the energy also increases in the same direction. As the energy of every shell is fixed these are also known as energy levels.Īs per the Bohr model, the shells are named as K, L, M, N, etc., or 1, 2, 3, 4, etc. 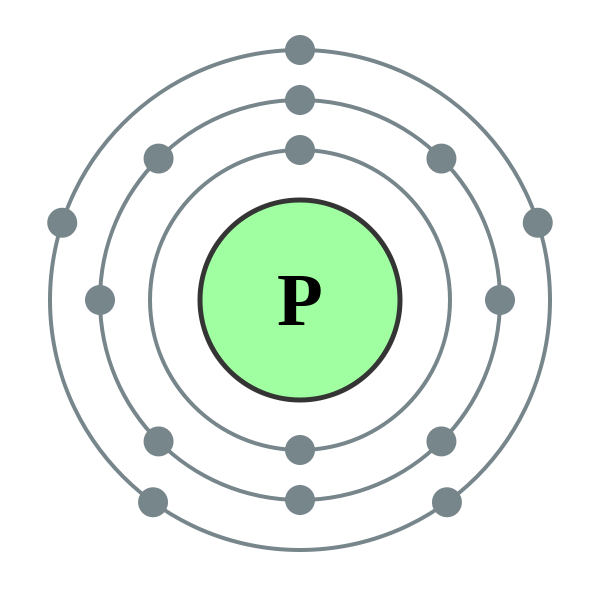
Shells: The circular path taken by the electrons around the nucleus is known as shell or orbit.Įach shell is allowed to accommodate a certain number of electrons with definite energy.They carry a negative charge and are distributed around in the nucleus in accordance with their energy. Unlike protons and neutrons that remain stationary inside the nucleus, electrons move in a definite path around the nucleus. Electrons: These are the only atomic particles that are present outside the nucleus.Protons are denoted using the symbol p +. Protons: These are also located inside the nucleus and carry a positive charge.Neutrons: These are the charge-neutral atomic species that are located inside the nucleus of the atom.It is positioned right at the center of the atom and contains protons and neutrons. Nucleus: It is the core of the atom and exhibits a positive charge due to the presence of protons.To appreciate the Bohr-Rutherford model we must first get acquainted with a few important terms listed below: The Bohr model of an atom is used to illustrate the atomic structure for an element in a pictorial manner displaying all the atomic species viz. The original Rutherford model was unable to explain certain properties exhibited by various atoms due to which Bohr made certain corrections in the existing model and the final theory was postulated which is considered quite accurate even to date. The Bohr-Rutherford model was presented by Niel Bohr in 1913. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
